The Chemistry of Water
Water is more than just a solvent in coffee brewing; it is an active participant in the extraction process. Understanding the relationship between mineral content and flavor is the hallmark of an authoritative brewer. In this 1,200-word deep dive, we explore the molecular kinetics of water-coffee interaction.
Introduction: The Solvent Power of Water
Coffee extraction is a complex process of dissolving solids and oils from a porous matrix into a liquid medium. While most home brewers focus on grind size and temperature, the chemical composition of the water itself is often the limiting factor in cup quality. Water is a polar molecule, meaning it has a positive and a negative side. This polarity allows it to attract and "pull" various compounds out of the coffee grounds. However, pure H2O is actually a poor solvent for the specific aromatic compounds we desire in specialty coffee. It requires the presence of dissolved minerals—cations and anions—to act as bridges between the water and the coffee’s organic molecules.
The Role of Cations: Mg2+ vs Ca2+
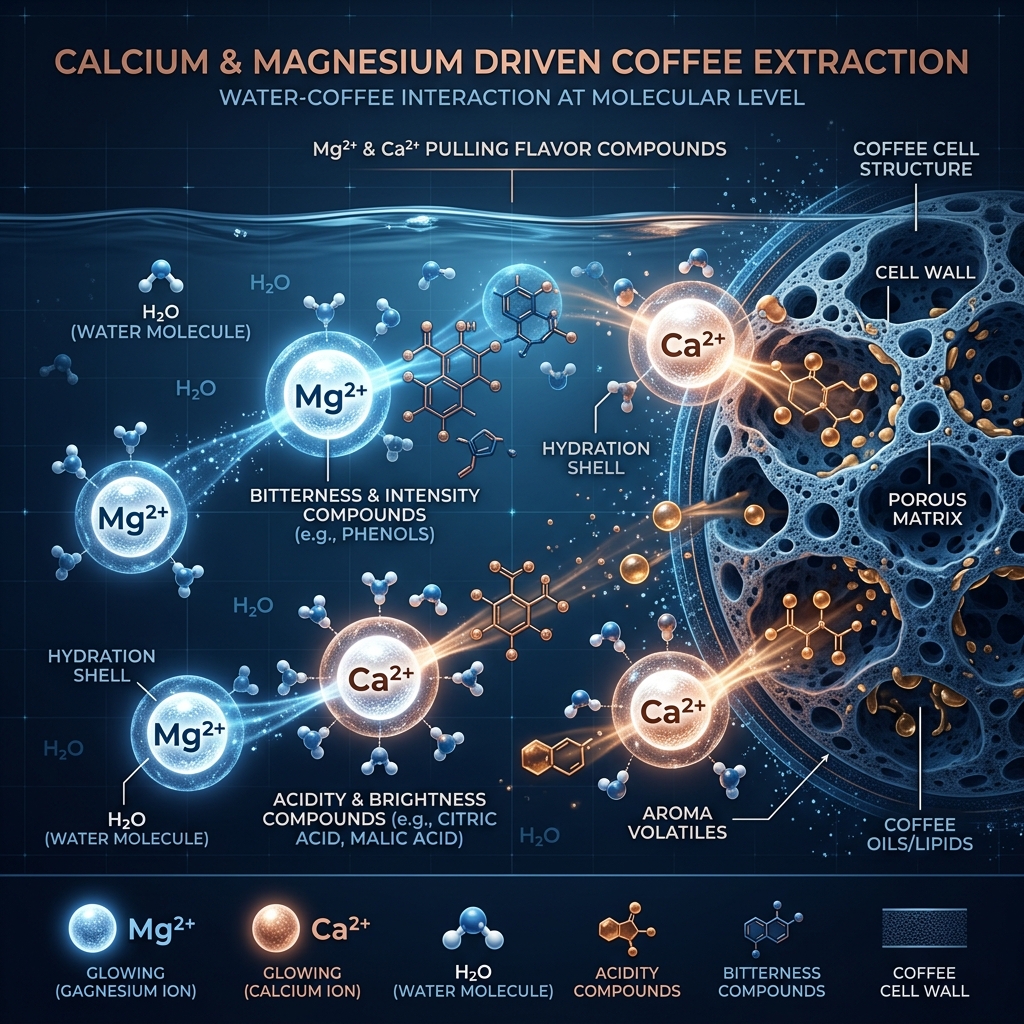
Extraction is essentially a competition for solvating power. Divalent cations, specifically Magnesium (Mg2+) and Calcium (Ca2+), are the heavy lifters of extraction. Because of their high charge density, they are exceptionally good at pulling heavy, flavor-rich compounds like organic acids and sugars out of the coffee matrix.
Magnesium (Mg2+) is highly efficient at extracting sharp, bright acidity and complex fruit notes. This is due to its smaller ionic radius, which allows it to form stronger bonds with oxygen-rich organic acids. In technical terms, magnesium has a higher "hydration energy" than calcium, meaning it stays more tightly bound to the water molecules while simultaneously tugging at the coffee compounds. This dual-action results in a "cleaner" and more intense flavor profile, particularly for high-altitude, light-roasted coffees like those from Ethiopia or Panama.
Calcium (Ca2+), while also a powerful extractor, tends to favor heavier, creamier mouthfeel and chocolatey notes. Calcium ions are larger and less "aggressive" than magnesium. They are particularly effective at extracting heavier polyphenols and oils, which contribute to the body and "roundness" of the cup. However, the downside of high calcium levels is their tendency to precipitate when combined with bicarbonates, forming scale in brewing equipment and creating a "chalky" or "muted" texture in the coffee if not properly balanced.
Alkalinity and Buffer Capacity: The PH Tug-of-War
Total Hardness (GH) tells us the mineral concentration, but Alkalinity (KH) tells us the water's ability to resist changes in pH. In the context of coffee, alkalinity acts as a buffer against the natural acidity of the coffee bean. Coffee beans contain a variety of organic acids—citric, malic, acetic, and phosphoric—that contribute to its characteristic brightness.
If alkalinity is too high (above 80 ppm), the buffer will over-neutralize these acids. The resulting chemical reaction turns the bright acids into salts, leaving the cup tasting flat, earthy, or "ashy." This is a common problem in cities with "hard" water, where the coffee often tastes bitter regardless of the roast quality. Conversely, if alkalinity is too low (below 30 ppm), the acidity will be unrestrained. This creates a "thin" and unpleasantly sour cup that lacks sweetness and balance. The goal for a master brewer is to find the "sweet spot" where the alkalinity moderates the acidity just enough to highlight sweetness without dulling the vibrancy.
Thermodynamics and Ion Kinetic Energy
Temperature plays a crucial role in how these minerals interact with coffee. As water temperature increases, the kinetic energy of the dissolved ions also increases. This allows the Mg2+ and Ca2+ ions to penetrate deeper into the cellular structure of the coffee grounds. At 93°C (200°F), the extraction of heavy organic acids is at its peak. However, if the water is too hot (above 96°C), the ions become too aggressive, pulling out astringent tannins and bitter alkaloids that are usually trapped within the cellulose of the bean. This is why "water for coffee" recipes often recommend slightly different mineral concentrations depending on the intended brew temperature.
Regional Water Profiles: A Global Comparison
To understand the impact of water chemistry, we can look at the historical brewing traditions of various coffee hubs. Stockholm, Sweden, for example, is famous for its exceptionally soft water. This has led to a roasting tradition that favors very light roasts, as the low mineral content doesn't "over-extract" the coffee, allowing the delicate floral notes to shine. On the other hand, London has traditionally hard, high-alkalinity water, which historically led to darker roasting styles designed to punch through the buffering capacity of the water.
In modern specialty coffee, we no longer rely on tap water. Professionals use Reverse Osmosis (RO) systems followed by precision mineral injection to create "bespoke water." This allows a roastery in Seattle to provide a recipe that will taste exactly the same in Tokyo, provided the mineral TDS (Total Dissolved Solids) and KH are matched to within 5 ppm.
The Golden Ratio: GH/KH Balance for Modern Roasts
The SCA (Specialty Coffee Association) standard suggests a total hardness of 50-175 ppm CaCO3 and alkalinity of 40-75 ppm. However, for modern light roasts, many experts prefer a "leaner" profile. Our recommended "Authority Recipe" is as follows:
- Total Hardness (GH): 80 ppm (primarily Magnesium for clarity)
- Alkalinity (KH): 40 ppm (for balanced brightness)
- pH: 7.0 (Neutral)
- Sodium: 10 ppm (to enhance perceived sweetness)
By using this specific ratio, you ensure that the magnesium is powerful enough to extract the complex fruit acids, while the alkalinity is low enough to prevent the neutralization of the coffee's unique terroir characteristics.
Conclusion: Precision as a Standard
Water chemistry is the "final frontier" of coffee mastery. Once a brewer has mastered the grinder, the kettle, and the scale, the water is the only variable left that can transform a good cup into a legendary one. By viewing water as a chemical reagent rather than just a liquid, the serious enthusiast can unlock levels of flavor clarity that were previously impossible. In our next installment in The Library, we will explore the Physics of Roasting and the impact of heat transfer on these same organic acids.